Hydrostatic Pressure
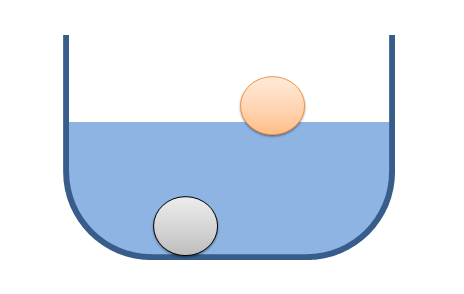
Hydrostatic pressure is the distributed force a liquid in equilibrium exerts on surfaces that are directly in contact with that fluid. If we imagine a bucket of water like the one shown below, the fluid (water in this case) exerts a force distributed over an area (a pressure) on all surfaces in contact with the water (the walls and bottom of the bucket, the entire surface of the metal ball, and the part of the surface of the foam ball that is in contact with the water.
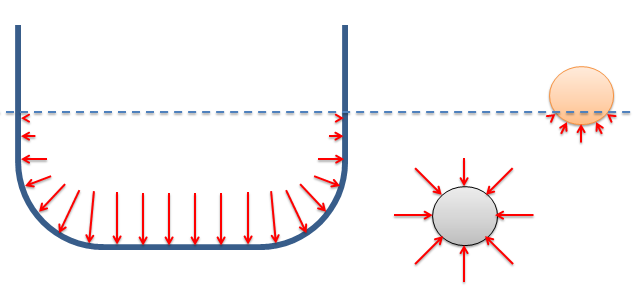
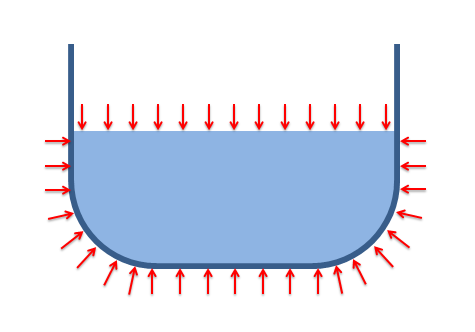
The magnitude of the pressure is equal to...
\[ \mathrm{ P = P_0 + \rho g h }\]Where \( \mathrm{P} \) is the pressure acting on the surface in contact with the liquid.
\( \mathrm{P_0} \) is the pressure at the surface of the liquid
\( \mathrm{\rho} \) is the density of the liquid (the density of water is 1000 kg/m^3)
\( \mathrm{g} \) is pull of gravity (9.81 N/kg on earth)
and \( \mathrm{h} \) is the depth of the fluid at the given location.
Hydrostatic pressures always act normal to the surface in contact with the fluid (fluids in equilibrium cannot exert frictional forces tangent to the surface).
Absolute versus Gage pressure:
The air we breathe and live in on earth also exerts a pressure on us, the ground, and the surfaces of liquids (hence the \( \mathrm{P_0} \) in the above equation). This atmospheric pressure at sea level is equal to 101.3 KPa (101300 N/m^2). Only in a perfect vacuum would objects experience no pressure acting on them. If we define a perfect vacuum as being zero pressure, we are using absolute pressure. To eliminate this atmospheric pressure acting on everything, engineers often use gage pressure instead. For gage pressure, we redefine standard atmospheric pressure as zero and vacuums become negative pressures. This usually simplifies analysis, since the atmosphere is in contact with most of the bodies we are examining and the atmospheric pressure cancels itself out anyway. It is usually safe to assume gage pressures, unless it is explicitly stated that absolute pressures are given.
 |
| Author: Jacob Moore has liscenced this work under a Creative Commons Attribution-NonCommercial 3.0 Unported License. |